 
For his work with radiation and the atomic nucleus, Rutherford received the 1908 Nobel Prize in chemistry. During the race in finding out the right model that could represent the atomic structure, many scientists came and the second amongst them was a. Acceptance of this model grew after it was modified with quantum theory by Niels Bohr. Below you will find a brief description on its theory and experiment along with the limitations that it faced. The conclusion is drawn that these chemical changes must be sub-atomic in character.\)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles. Rutherford shared his insightful knowledge on the atomic model. “Radioactivity is shown to be accompanied by chemical changes in which new types of matter are being continually produced. “All science is either physics or stamp collecting.” This meant that an electron circling the nucleus. But the Rutherford atomic model used classical physics and not quantum mechanics. Far from the nucleus are the negatively charged electrons. Most of the mass is in the nucleus, and the nucleus is positively charged. Rutherford was laid to rest in Westminster Abbey, the same location as Charles Darwin and Isaac Newton.įamous Ernest Rutherford quotes include: “If you can't explain your physics to a barmaid it is probably not very good physics.” The Rutherford atomic model was correct in that the atom is mostly empty space. It is a synthetic, radioactive element made in particle accelerators. 
The element 'rutherfordium' (atomic number 104) was named in Rutherford’s honor. In 1908, Rutherford was awarded the Nobel Prize in Chemistry for his work on the transmutation of elements and the chemistry of radioactive material. 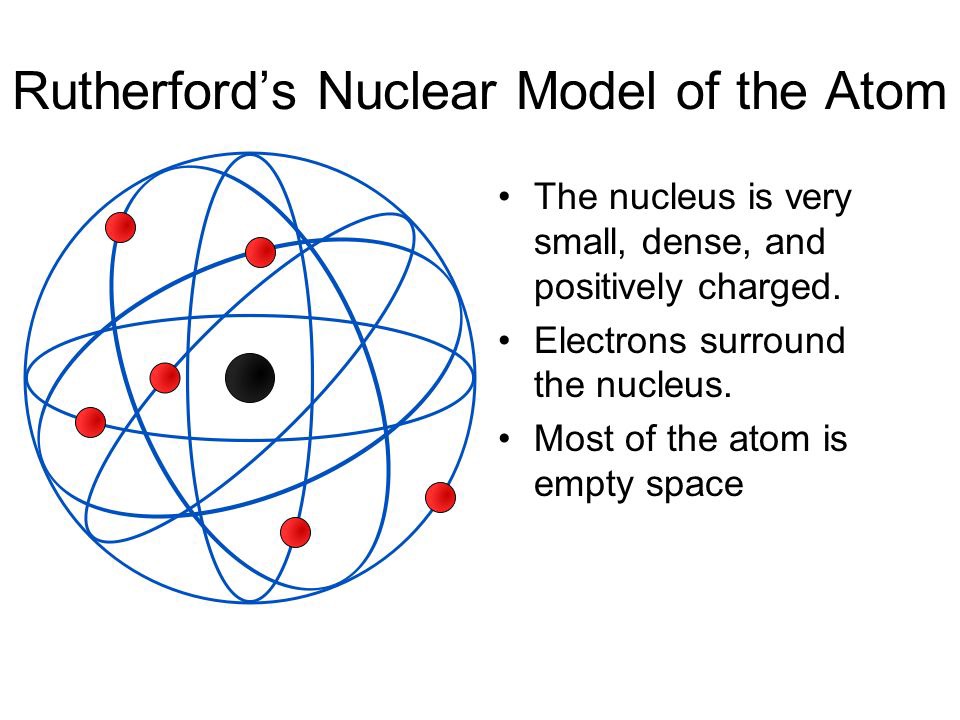
This symbol became popular and has been used by various organizations around the world as a symbol for atoms and atomic energy in general. The Rutherford model of the atom was simplified in a well known symbol showing electrons circling around the nucleus like planets orbiting the sun. Thomson, Ernest Rutherford, Niels Bohr, Robert Millikan. The surprising results of this experiment demonstrated the existence of the atomic nucleus and became an integral part of the Rutherford model of the atom. Who are the main scientists involved in atomic theory The main scientists involved in early atomic theory are Democritus, John Dalton, J.J. In 1907, Rutherford, Hans Geiger and Ernest Marsden carried out the Geiger-Marsden experiment, an attempt to examine the structure of the atom. Ernest Rutherford was a New Zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom Rutherford model is also known as the Rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Physicist Ernest Rutherford envisioned the atom as a miniature solar system, with electrons orbiting around a massive nucleus, and as mostly empty. He performed experiments on radioactivity and is widely regarded as the Father of Nuclear Physics or Father of the Nuclear Age. Atomic model, in physics, a model used to describe the structure and makeup of an atom. Ernest Rutherford was the first man to split an atom, transmuting one element into another. He also observed that radioactive material took the same amount of time for half of it to decay, known as its “half life”. For a more in-depth discussion of the history of atomic models, see atom: development of atomic theory. 
Rutherford worked on radioactivity, coining the terms ‘alpha’ and ‘beta’ to describe the two different types of radiation emitted by uranium and thorium. Rutherford studied at Canterbury College, University of New Zealand before moving to England in 1895 for post graduate study at Cavendish Laboratory, University of Cambridge. Ernest Rutherford lived from the 30th of August 1871 to the 19th of October 1937. Rutherfords Atomic Model is based on the observations of -particles bombarding a gold foil and the deflection of these particles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
